18072885002
Ceramic membranes have emerged as transformative separation and filtration tools in the biotechnology and food processing industries. Their unique physicochemical properties — high thermal stability, chemical resistance, and tunable pore structures — make them especially suited for the demanding conditions encountered in collagen protein production. This article reviews the applications of ceramic membranes across the collagen manufacturing workflow, from raw material pre-treatment to downstream purification, and evaluates their performance relative to conventional polymeric membranes. Key challenges and emerging innovations are also discussed.
Ceramic UF membranes with molecular weight cut-offs (MWCO) of 1–50 kDa provide reproducible size-based fractionation of collagen hydrolysates. Unlike polymeric membranes, ceramic UF membranes maintain their pore structure integrity even after repeated exposure to the proteolytic enzyme solutions used in hydrolysis, and can be thoroughly regenerated with hot sodium hydroxide and nitric acid without pore enlargement or membrane compression.
A typical fractionation scheme might involve sequential passes through 10 kDa and 3 kDa ceramic UF membranes to produce enriched peptide fractions with defined molecular weight profiles for specific functional food or pharmaceutical applications.
Applications of Ceramic Membranes in Collagen Processing
1. Pre-treatment and Clarification of Extraction Broths
One of the earliest and most critical applications of ceramic membranes in collagen production is the clarification of raw extraction broths. After extraction or enzymatic hydrolysis, the process liquor contains a heterogeneous mixture of soluble proteins, lipid emulsions, colloidal particles, and residual suspended solids.
Microfiltration ceramic membranes with pore sizes of 0.1–0.5 µm are ideally suited for this clarification step. They efficiently remove turbidity-causing particles, fat globules, and microbial contaminants while allowing collagen peptides and smaller protein fragments to permeate freely. The ability to operate ceramic MF membranes in cross-flow mode at elevated temperatures (50–70°C) — conditions that prevent microbial growth and reduce viscosity — is a significant operational advantage over polymeric systems.
Studies using alumina and zirconia microfiltration membranes have demonstrated high flux recovery and low protein retention losses during clarification of fish collagen hydrolysates, achieving turbidity reductions of greater than 99% while maintaining collagen peptide yields exceeding 90%.
2. Fractionation of Collagen Peptides by Molecular Weight
The bioactivity of collagen peptides is strongly influenced by their molecular weight. Peptides in the range of 1–5 kDa are commonly associated with enhanced bioavailability and specific biological effects such as stimulation of fibroblast collagen synthesis and antihypertensive activity. Achieving precise molecular weight cut-offs requires carefully controlled ultrafiltration.
Ceramic UF membranes with molecular weight cut-offs (MWCO) of 1–50 kDa provide reproducible size-based fractionation of collagen hydrolysates. Unlike polymeric membranes, ceramic UF membranes maintain their pore structure integrity even after repeated exposure to the proteolytic enzyme solutions used in hydrolysis, and can be thoroughly regenerated with hot sodium hydroxide and nitric acid without pore enlargement or membrane compression.
A typical fractionation scheme might involve sequential passes through 10 kDa and 3 kDa ceramic UF membranes to produce enriched peptide fractions with defined molecular weight profiles for specific functional food or pharmaceutical applications.
3. Concentration of Collagen Solutions
Before drying, dilute collagen or collagen peptide solutions must be concentrated to reduce energy costs in downstream spray drying or freeze drying. Ceramic UF membranes with appropriate MWCO can concentrate collagen solutions from initial concentrations of 1–3% (w/v) to 15–25% (w/v) while simultaneously removing low-molecular-weight impurities such as salt ions and small organic molecules.
The high mechanical stability of ceramic membranes allows operation at elevated transmembrane pressures (up to 10 bar) without risk of membrane compaction — a common limitation of polymeric membranes under high-pressure conditions. This enables efficient concentration in fewer process cycles, reducing processing time and energy consumption.
4. Diafiltration for Desalting
Collagen extraction and hydrolysis processes frequently use acidic or saline buffer systems that must be removed prior to final product formulation. Diafiltration — the continuous addition of water or buffer to a retained protein fraction while simultaneously filtering out low-molecular-weight solutes — is highly effective for desalting.
Ceramic membranes are particularly advantageous for diafiltration because their chemical stability allows sequential diafiltration cycles with water, followed by CIP with sodium hydroxide, without membrane degradation. Research has shown that ceramic membrane diafiltration of collagen hydrolysates can reduce salt content (measured as ash content) from 8–12% to less than 1% in a single diafiltration sequence, meeting requirements for food-grade specifications.
5. Removal of Pigments and Non-collagenous Proteins
Raw collagen extracts, particularly from bovine and porcine sources, often contain pigmented compounds (e.g., heme-derived molecules from residual blood) and non-collagenous proteins that impair product quality. Ceramic nanofiltration membranes, combined with activated carbon pre-treatment, can selectively remove these contaminants while retaining collagen peptides.
The stable hydrophilic surface of alumina and titania ceramic membranes resists irreversible adsorption of colored compounds, facilitating effective cleaning and maintaining flux over extended operation — a known challenge with polymeric NF membranes that can become permanently stained by proteinaceous pigments.
6. Sterile Filtration for Pharmaceutical-Grade Collagen
For collagen intended for pharmaceutical or biomedical applications — including injectable collagen for dermal filling, collagen scaffolds for tissue engineering, or collagen-based hemostatic agents — sterility is a non-negotiable requirement. Ceramic membranes with 0.2 µm pore size are effective for terminal sterilizing filtration, removing bacteria and particulates while preserving the integrity of collagen macromolecules.
The ability to autoclave ceramic membrane modules without pore alteration or structural failure gives them a critical advantage in pharmaceutical manufacturing environments where repeated sterilization cycles are standard practice.
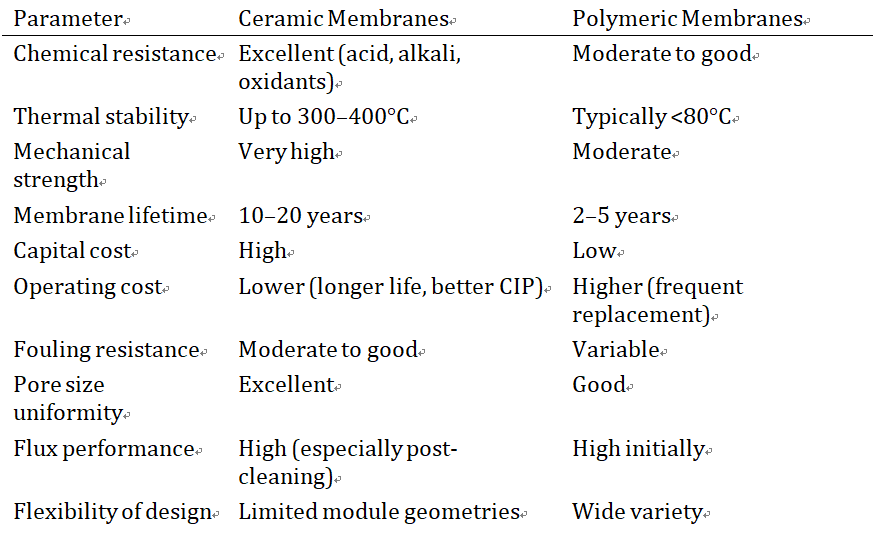
Comparison with Polymeric Membranes
For collagen processing, where repeated exposure to acidic and alkaline conditions is inherent and product hygiene standards are stringent, the total cost of ownership of ceramic membranes often compares favorably over multi-year operational horizons, despite higher initial capital investment.
Emerging Developments and Future Directions
1. Surface-Modified Ceramic Membranes
Researchers are investigating the functionalization of ceramic membrane surfaces with hydrophilic polymers (e.g., polyethylene glycol), zwitterionic coatings, or silane-based groups to further reduce protein adsorption and improve anti-fouling performance in collagen-rich environments. Such surface modifications aim to combine the structural advantages of ceramic substrates with the low-fouling surfaces achieved by specialized polymeric membranes.
2. Silicon Carbide (SiC) Membranes
Silicon carbide membranes represent a next-generation ceramic membrane material with exceptional hydraulic permeability, superior chemical resistance, and outstanding hydrophilicity compared to alumina- and titania-based membranes. Early industrial applications suggest that SiC membranes can achieve significantly higher fluxes in protein processing streams, potentially reducing membrane area requirements and capital costs in new installations.
3. Hybrid Membrane-Enzyme Systems
The integration of immobilized collagenolytic enzymes within ceramic membrane modules — so-called membrane bioreactors — is an area of active research. In such systems, enzymatic hydrolysis and simultaneous separation of the resulting peptides occur within a single unit operation, reducing processing time, minimizing thermal denaturation, and enabling continuous rather than batch processing of collagen.
4. Process Intensification and Continuous Manufacturing
The collagen industry, like the broader biopharmaceutical sector, is moving toward continuous processing paradigms to improve productivity and reduce operational variability. Ceramic membranes — with their durability and tolerance for continuous operation over extended periods — are well-positioned to serve as key unit operations in continuous collagen purification trains, integrating with continuous extraction and enzymatic hydrolysis systems.
5. Regulatory and Quality Considerations
For collagen products intended for food, nutraceutical, pharmaceutical, or medical device applications, regulatory compliance is paramount. Ceramic membranes offer advantages in this context: their inorganic composition means there is no risk of leaching organic plasticizers or monomers into the product stream — a concern associated with certain polymeric membrane materials. Their extractable and leachable profiles are well-characterized and generally benign.
Ceramic membrane systems used in pharmaceutical collagen production must comply with Good Manufacturing Practice (GMP) requirements, including validated cleaning procedures, documented material compatibility, and traceability of membrane components. The well-defined pore structure and chemical stability of ceramic membranes facilitate the development and validation of reproducible cleaning and integrity testing protocols.
Conclusion
Ceramic membranes have established themselves as valuable tools across the collagen protein production workflow, from initial clarification of extraction broths through to the precise fractionation, concentration, desalting, and sterile filtration of final products. Their unmatched chemical and thermal stability, combined with long operational lifetimes and compatibility with aggressive cleaning regimens, make them particularly well-suited to the demanding processing environments inherent in collagen manufacturing.
While capital costs remain higher than polymeric alternatives, the total cost of ownership — considering membrane longevity, cleaning efficiency, and process reliability — increasingly favors ceramic membranes for industrial-scale collagen production. Advances in surface modification, novel materials such as silicon carbide, and the integration of ceramic membranes into continuous and intensified processing systems promise to further expand their role and improve their performance in this growing field.
As global demand for high-purity, bioactive collagen products continues to rise, ceramic membrane technology will play an increasingly central role in enabling manufacturers to meet the twin imperatives of process efficiency and product quality.